Currently Empty: $0.00
Packaging Type: 1 Vial
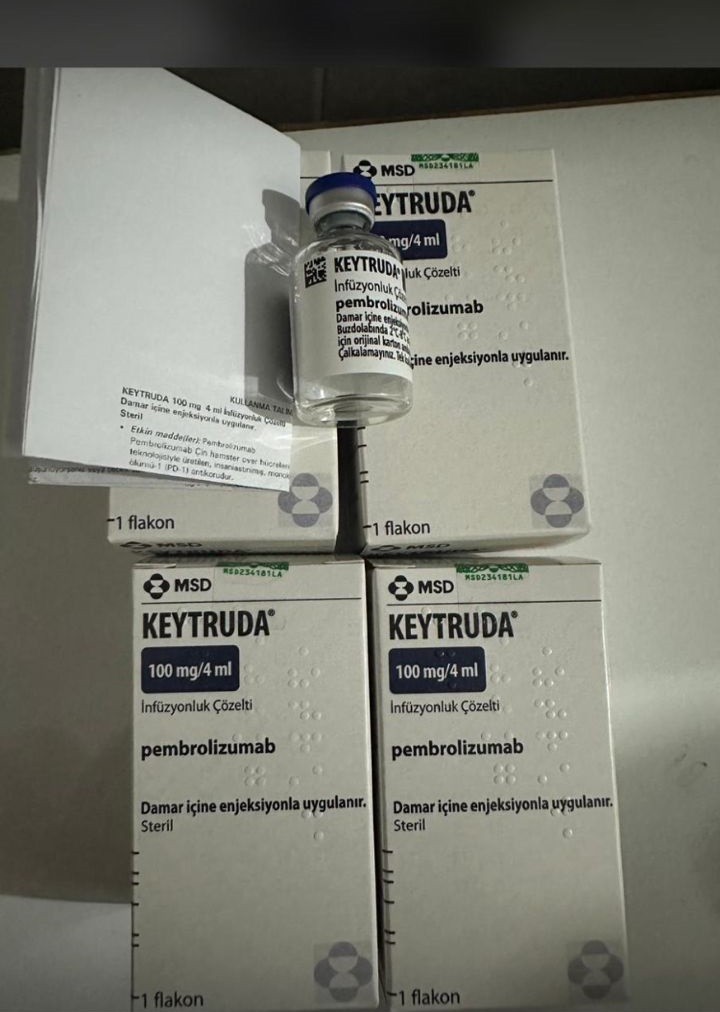
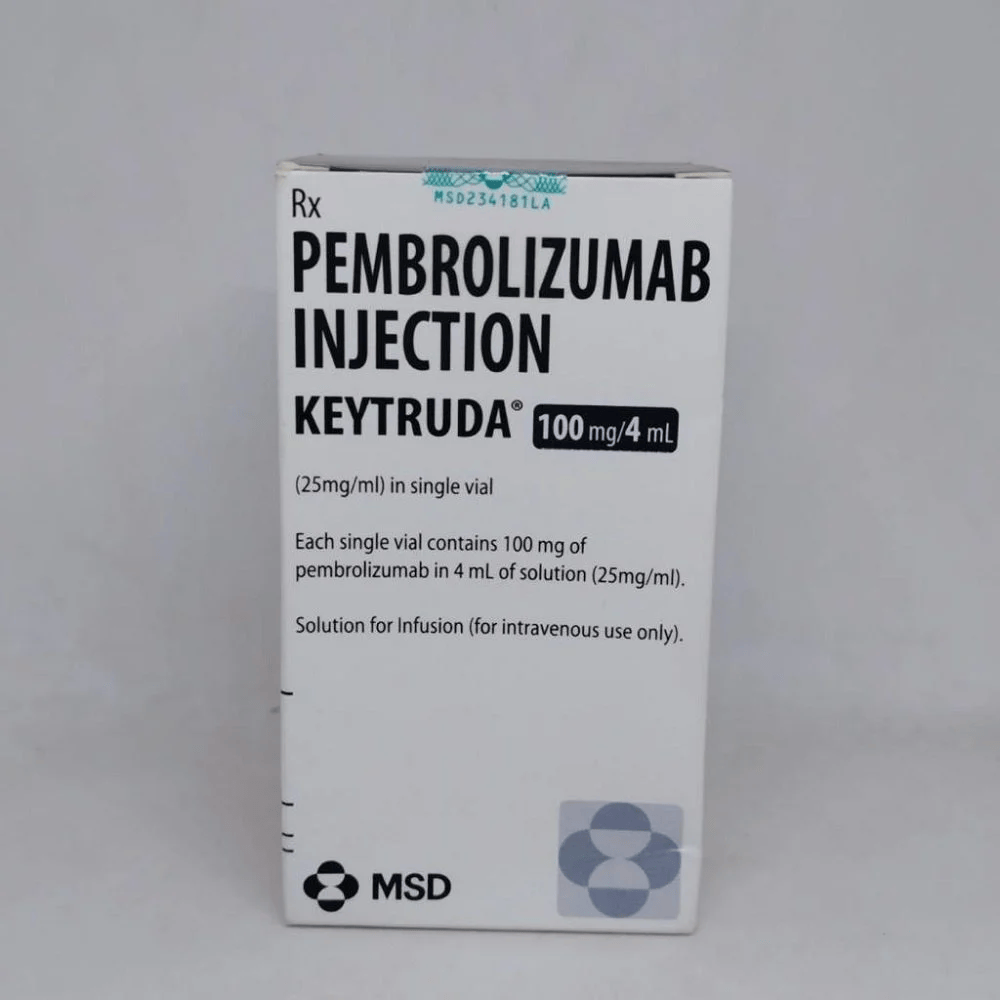
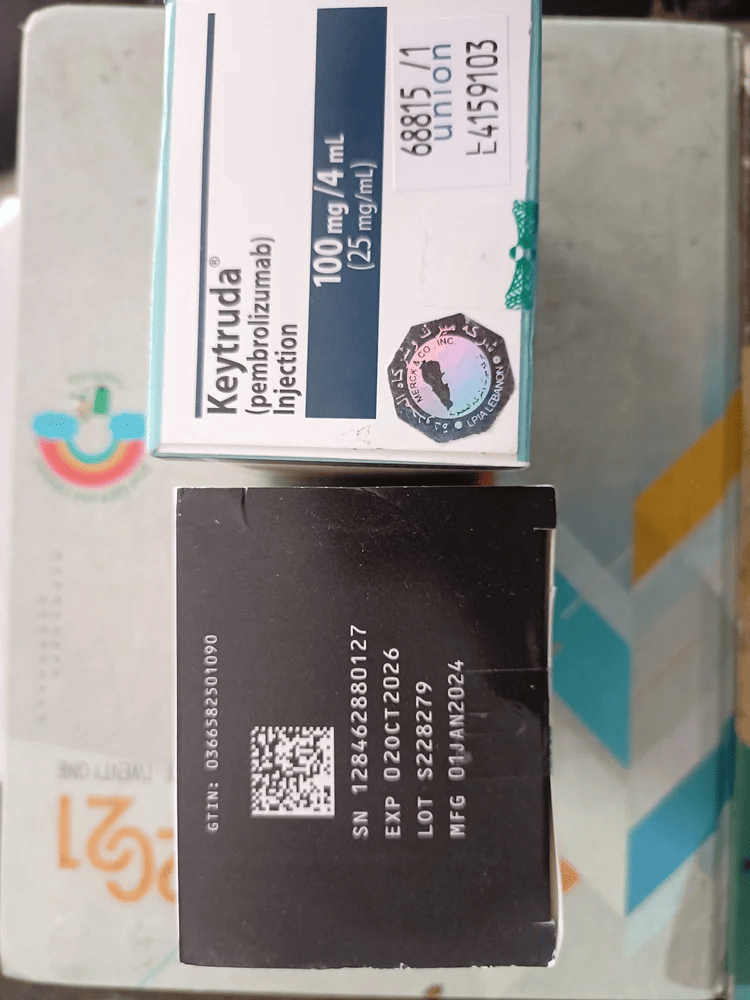
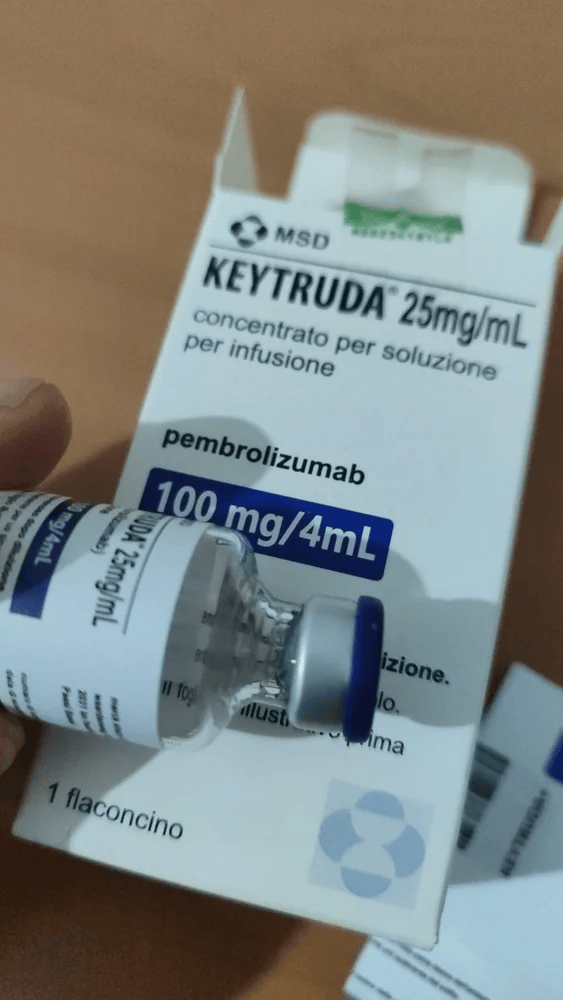
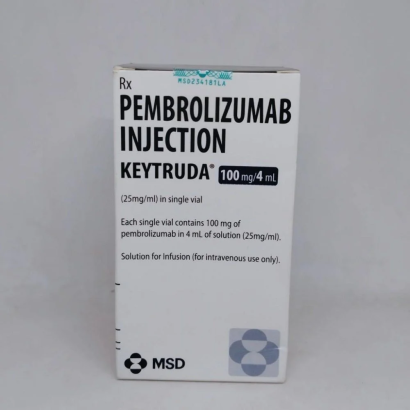
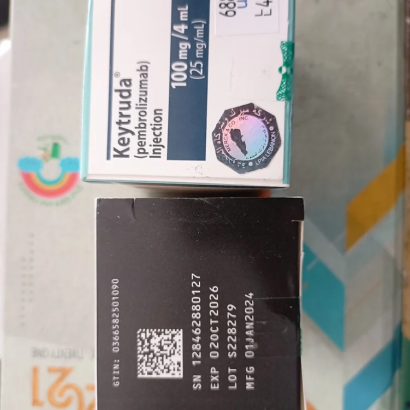
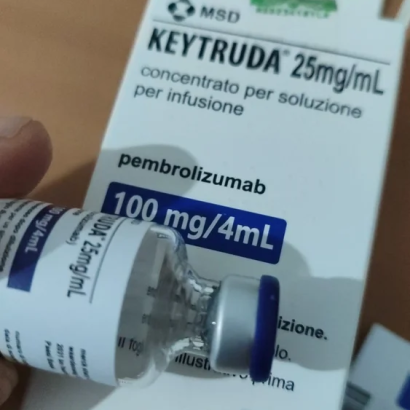
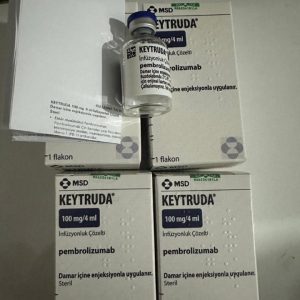
Composition: Pembrolizumab 100mg/4ml
Usage: Personal
Treatment: cancer called head and neck squamous cell cancer (HNSCC)
Strength: 100 mg
Shelf Life: 720
Item Weight: 30 g
SIZE GUIDE
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 88 | 89 | 90 | 91 | 92 | 93 |
| WAIST | 55 | 56 | 58 | 59 | 62 | 65 |
| HIPS | 90 | 92 | 94 | 95 | 96 | 98 |
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 60 | 62 | 65 | 66 | 68 | 69 |
| WAIST | 50 | 55 | 58 | 62 | 65 | 66 |
| HIPS | 80 | 82 | 85 | 86 | 88 | 89 |
| Size | US | UK / AUS | France | Italy | Japan | Denmark |
|---|---|---|---|---|---|---|
| XS | 0-2 | 6 | 34 | 38 | 5 | 32 |
| S | 2-4 | 8 | 36 | 40 | 7 | 34 |
| M | 6-8 | 10 | 38 | 42 | 9 | 36 |
| L | 8-10 | 12 | 40 | 44 | 11 | 38 |
Description
About Keytruda 100 mg Injection 4 ml
Keytruda 100 mg Injection 4 ml is an anti-cancer medicine used in the treatment of various types of cancers in both adult and paediatric populations. Cancer is a disease caused due to the abnormal growth of cells that divide uncontrollably. This medicine is used in treating advanced renal cell carcinoma, hepatocellular carcinoma, colorectal cancer, non-small cell lung cancer, metastatic melanoma, small cell lung cancer, gastric cancer, classical Hodgkin’s disease, urothelial carcinoma, cervical cancer, primary mediastinal large B-cell lymphoma, endometrial cancer, microsatellite instability-high cancer, and head and neck cancer.
Keytruda 100 mg Injection 4 ml contains Pembrolizumab, which belongs to the class of monoclonal antibodies. It works by inhibiting the action of a programmed death receptor-1 (PD-1) and thereby stops the abnormal growth of cancer cells.
Keytruda 100 mg Injection 4 ml may cause certain side effects such as diarrhoea, nausea, vomiting, tiredness, itching, rash, headache, fever, cough, shortness of breath, muscle/joint pain, loss of appetite and injection site reactions. Inform your doctor if any of these side effects persist for a longer duration. Keytruda 100 mg Injection 4 ml will be administered by an oncologist. Avoid self-administration.
Avoid taking Keytruda 100 mg Injection 4 ml if you are allergic to it or any other components of it. Before taking Keytruda 100 mg Injection 4 ml, inform your doctor if you have/had liver or kidney disease, autoimmune disorders such as ulcerative colitis, Crohn’s disease, any history of organ transplants, received a stem cell transplant that uses donor stem cells (allogeneic), undergone radiation treatment to the chest or any other medical conditions. This medicine is known to cause embryo-fetal toxicity. Hence, it is not recommended for use in pregnancy and breastfeeding.
Uses of Keytruda 100 mg Injection 4 ml
Directions for Use
Medicinal Benefits
Keytruda 100 mg Injection 4 ml contains Pembrolizumab, which is used in the treatment different types of cancers. It works by inhibiting the action of a programmed death receptor-1 (PD-1) that causes the multiplication of cancerous cells. This helps stop the abnormal growth of cancer cells and thus reduce the further spread to other parts of the body.
How Keytruda 100 mg Injection 4 ml Works
Storage
Side Effects of Keytruda 100 mg Injection 4 ml
- Nausea
- Vomiting
- Diarrhoea
- Loss of appetite
- Tiredness
- Dizziness
- Weakness
- Pain in muscles/joints/bones
- Cough
- Headache
- Fever
- Itching
- Rash
- Back pain
- Shortness of breath
- Injection site reactions
What if I have taken an overdose of Keytruda 100 mg Injection 4 ml
Drug Warnings
To treat your condition effectually, continue taking Keytruda 100 mg Injection 4 ml for the period that your doctor has prescribed. Keytruda 100 mg Injection 4 ml may cause severe immune-mediated adverse reactions (IMAR) such as immune-mediated colitis, hepatitis (hepatotoxicity), nephritis, endocrinopathies (such as adrenal insufficiency, diabetes, thyroid disorders), pneumonitis, and dermatologic reactions (such as Stevens-Johnson syndrome, epidermal necrolysis). It may also cause infusion-related reactions in some patients. Therefore, caution should be exercised if you have/had liver or kidney disease, any history of organ transplant, received a stem cell transplant that uses donor stem cells (allogeneic), undergone radiation treatment to the chest and autoimmune disorders such as ulcerative colitis, or Crohn’s disease while receiving Keytruda 100 mg Injection 4 ml. Discontinue the treatment if any severe infusion reactions occur. Inform your doctor if you are pregnant or are planning to become pregnant before starting the treatment, as this medicine is known to cause embryo-fetal toxicity. Breastfeeding should be discontinued in nursing mothers during the treatment as it passes into breast milk and may cause a possible risk to the infant. This medicine is not recommended for use in children below 2 years of age as the safety and efficacy are not established.
Diet & Lifestyle Advise
- Physical activity helps strengthen muscles, reduces fatigue, helps in weight loss, and gives strength. Gentle activities like 20-30 minutes of walking or swimming would be helpful.
- Maintain a healthy weight by performing regular low-strain exercises and eating healthy food.
- Getting adequate sleep as resting helps improve your health and mental ability and improves attention.
- De-stress yourself by meditating, reading books, taking a warm bubble bath, or listening to soothing music.
- Eat food rich in antioxidants such as berries, spinach, etc.
- Foods containing fibre help in better digestion. These include beans, peas, lentils, whole grains, nuts, and seeds.
- Avoid smoking and alcohol consumption.
- Maintain a healthy weight.
Habit Forming
Therapeutic Class
Additional information
| Qty | 100mg/Vial |
|---|














There are no reviews yet.