Currently Empty: $0.00
Packaging Size: pack of 20 tablets
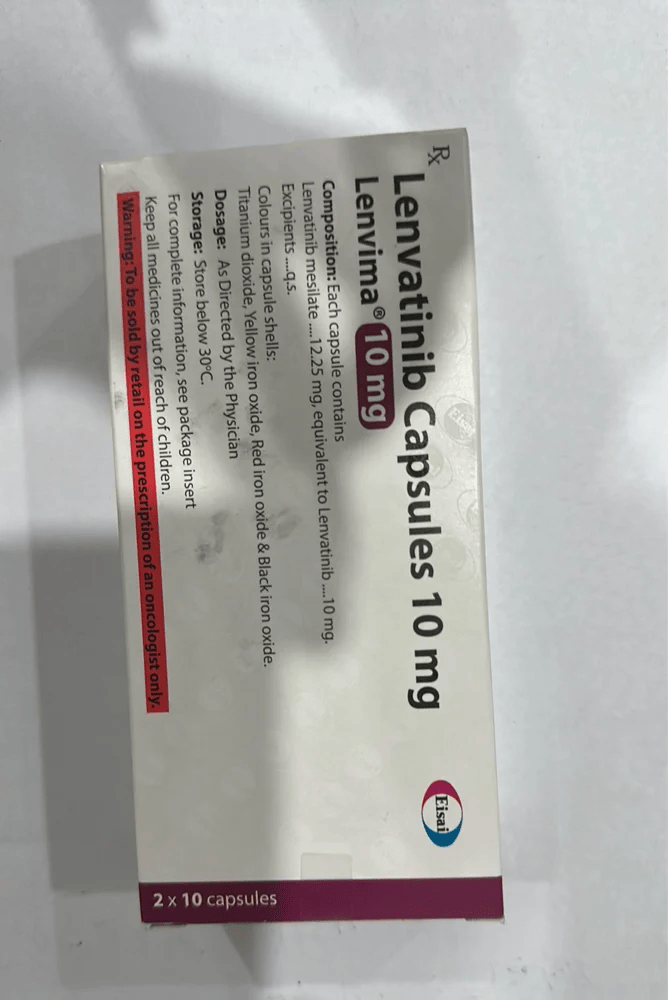
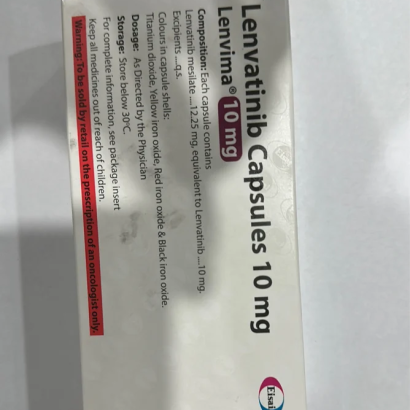
Strength: 4mg/10mg/12mg/18mg/24mg
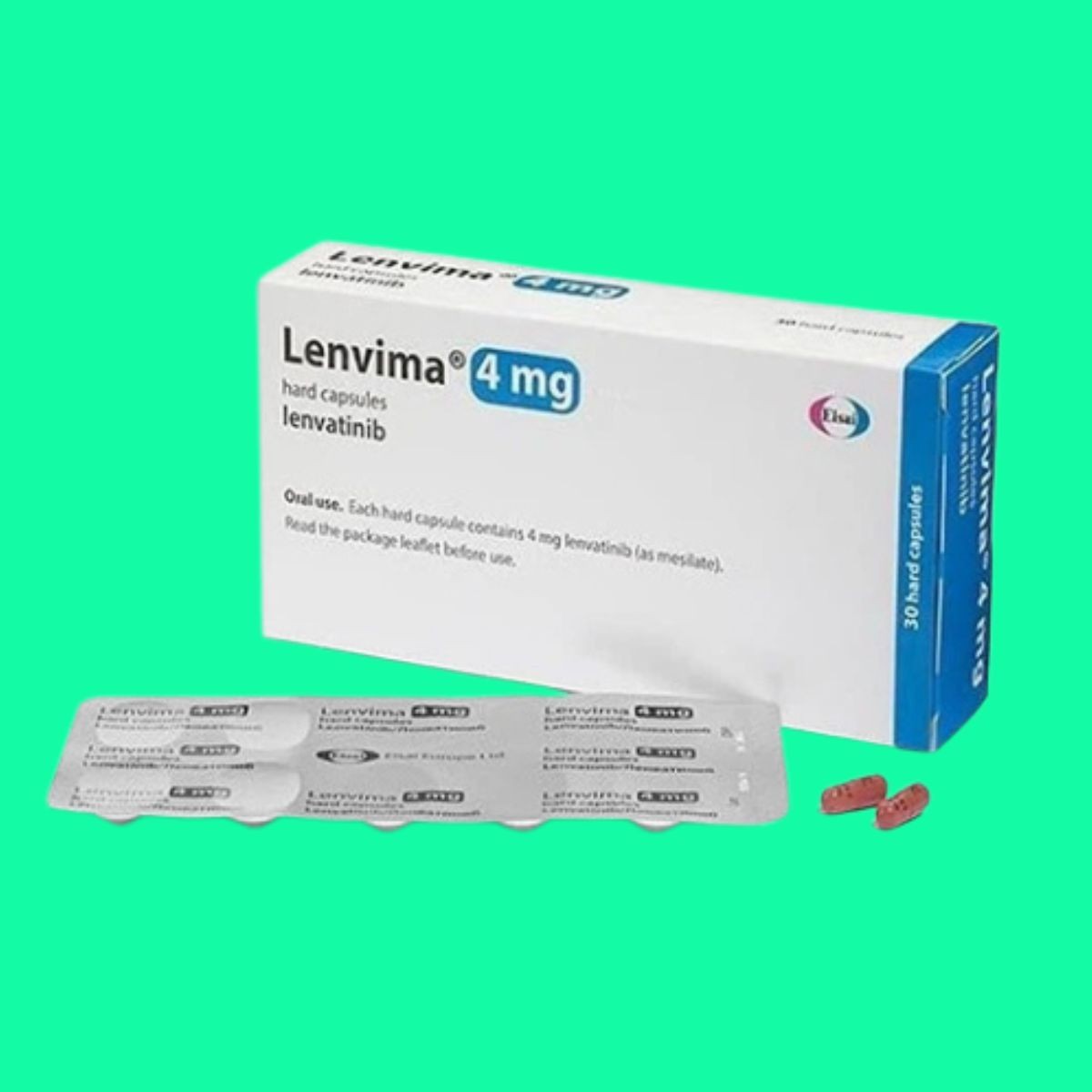
Composition: Lenvatinib (4mg)
Treatment: Thyroid cancer
Prescription/Nonprescription: Prescription
Form: Tablet
Shelf life: 3 years
Minimum Order Quantity: 1 Box
Uses of Lenvima 4mg Capsule
Lenvima 4mg Capsule is indicated to treat the following conditions:
- Certain types of advanced thyroid cancer
- Advanced Liver cancer
- Certain types of kidney cancer
- Endometrial cancer
SIZE GUIDE
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 88 | 89 | 90 | 91 | 92 | 93 |
| WAIST | 55 | 56 | 58 | 59 | 62 | 65 |
| HIPS | 90 | 92 | 94 | 95 | 96 | 98 |
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 60 | 62 | 65 | 66 | 68 | 69 |
| WAIST | 50 | 55 | 58 | 62 | 65 | 66 |
| HIPS | 80 | 82 | 85 | 86 | 88 | 89 |
| Size | US | UK / AUS | France | Italy | Japan | Denmark |
|---|---|---|---|---|---|---|
| XS | 0-2 | 6 | 34 | 38 | 5 | 32 |
| S | 2-4 | 8 | 36 | 40 | 7 | 34 |
| M | 6-8 | 10 | 38 | 42 | 9 | 36 |
| L | 8-10 | 12 | 40 | 44 | 11 | 38 |
Description
What is Lenvima (lenvatinib) for?
Lenvima (lenvatinib) is a kinase inhibitor (targeted therapy) indicated to treat:
- For the first-line treatment of patients with a type of liver cancer called hepatocellular carcinoma (HCC) when it cannot be removed by surgery.
- For the first-line treatment of adults with a type of thyroid cancer called differentiated thyroid cancer (DTC), that can no longer be treated with radioactive iodine and is progressing.
- In combination with everolimus, for the treatment of patients with advanced renal cell carcinoma (RCC), a type of kidney cancer, after one course of treatment with another anti-cancer therapy.
It is available in capsule form, containing 4 mg or 10 mg lenvatinib.
How does Lenvima (lenvatinib) work?
Lenvatinib is a targeted treatment. It is a multiple receptor tyrosine kinase inhibitor that targets certain proteins on cancer cells that cause the cells to grow and multiply. It selectively inhibits the activities of the proteins VEGFR, FGFR, RET, KIT and PDGFR.
Lenvatinib is thought to block the signals that allow cells (cancer and normal) to survive and multiply. It is thought to block signals that help blood vessels grow. Blood vessels support the tumor’s survival and growth.
Because normal cells also have these proteins, they may be affected by treatment.
Where has Lenvima (lenvatinib) been approved?
Currently, Lenvima (lenvatinib) has been approved for the treatment of:
- Differentiated thyroid cancer in over 70 countries including Japan, in Europe, China and in Asia, and in the United States for radioiodine-refractory differentiated thyroid cancer.
- Inoperable liver cancer in over 65 countries including Japan, the United States, in Europe, China and in Asia.
- Advanced kidney cancer in combination with everolimus following prior antiangiogenic therapy in over 60 countries, including the United States, in Europe and in Asia. In Europe, the medicine was launched under the brand name Kisplyx® for kidney cancer.
- Endometrial cancer in combination with KEYTRUDA, for cancer that is not microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), who have disease progression following prior systemic therapy and are not candidates for curative surgery or radiation in over 10 countries including the United States, Canada and Australia.
- Unresectable thymic cancer in Japan.
It is also being studied for the treatment of other types of cancer
Please note that this medicine may have also been approved in other regions than the ones we’ve listed. If you have a question about its approval in a specific country feel free to
How is Lenvima (lenvatinib) taken?
The standard dosage is
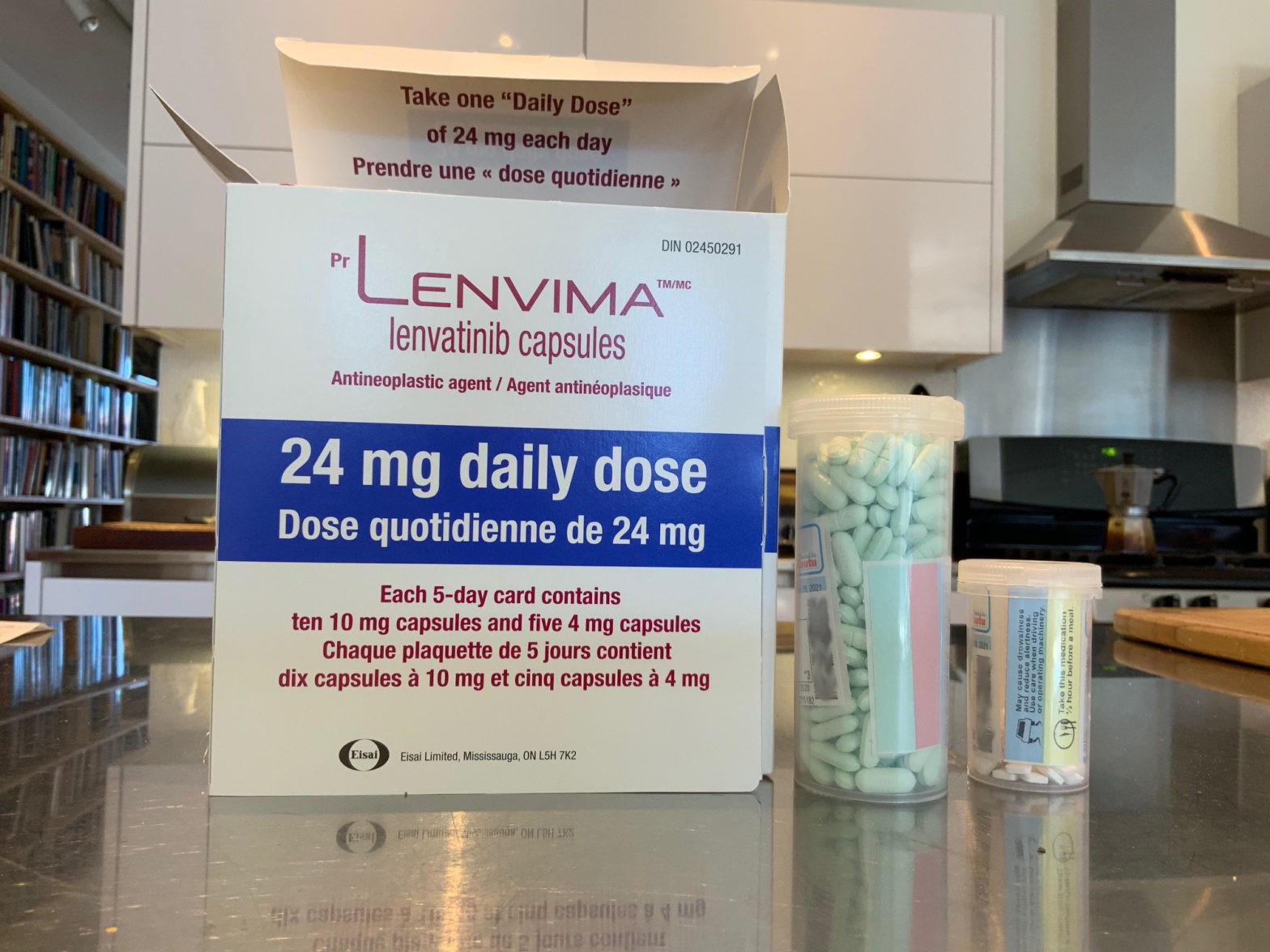
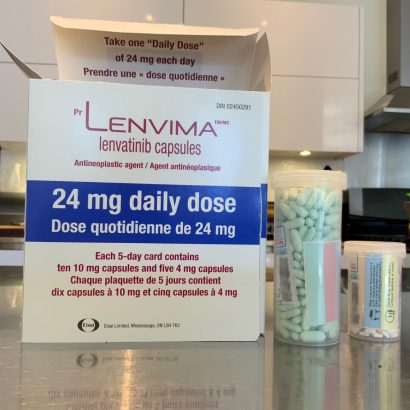
- DTC: 24 mg (two 10 mg capsules and one 4 mg capsule) orally once daily.
- RCC: 18 mg orally once daily with everolimus 5 mg orally once daily.
- HCC: based on actual body weight:
- for patients greater than or equal to 60 kg: 12 mg (three 4 mg capsules) orally once daily
- for patients less than 60 kg: 8 mg (two 4 mg capsules) orally once daily
The dose should be taken at around the same time, with or without food. The capsules should be swallowed whole with water.
The recommended daily dose should be modified for certain patients with kidney or liver problems.
Complete information about Lenvima (lenvatinib) dosage and administration can be found in the official prescribing information listed in our references section.
Note: Please consult with your treating doctor for personalised dosing.
Are there any known adverse reactions or side effects of Lenvima (lenvatinib)?
Common adverse reactions
The most common side effects ( ≥10% of patients) listed in the prescribing information include:
- tiredness
- joint and muscle pain
- decreased appetite
- weight loss
- nausea
- mouth sores
- headache
- vomiting
- rash
- redness
- itching
- peeling of your skin on your hands and feet
- stomach (abdomen) pain
- hoarseness
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:
- high blood pressure (hypertension)
- heart problems
- problem with blood clots in your blood vessels (arteries)
- liver problems
- kidney problems
- increased protein in your urine (proteinuria)
- diarrhea
- an opening in the wall of your stomach or intestines (perforation) or an abnormal connection between two or more body parts (fistula)
- changes in the electrical activity of your heart called QT prolongation
- low levels of blood calcium (hypocalcemia)
- a condition called Reversible Posterior Leukoencephalopathy Syndrome (RPLS)
- bleeding
- change in thyroid hormone levels
- wound healing problems
- severe jaw bone problems (osteonecrosis)
Use in a specific population
Lenvima (lenvatinib) can be fatal for a fetus, it is advised to avoid pregnancies and breastfeeding. This medicine may cause fertility problems in males and females. Talk to your treating doctor if this is a concern for you.[1]
For a comprehensive list of side effects and adverse reactions please refer to the official prescribing information.


















There are no reviews yet.