Currently Empty: $0.00
Form: Injection
Usage/Application: Hospital
Prescription/Nonprescription
Strength:50 mg
Minimum Order Quantity:01 Box
Afatinib, sold under the brand name Gilotrif among others, is a medication which is used to treat non-small cell lung carcinoma. It belongs to the tyrosine kinase inhibitor family of medications. It is taken by mouth
SIZE GUIDE
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 88 | 89 | 90 | 91 | 92 | 93 |
| WAIST | 55 | 56 | 58 | 59 | 62 | 65 |
| HIPS | 90 | 92 | 94 | 95 | 96 | 98 |
| SIZE | XS | S | M | L | XL | XXL |
|---|---|---|---|---|---|---|
| BUST | 60 | 62 | 65 | 66 | 68 | 69 |
| WAIST | 50 | 55 | 58 | 62 | 65 | 66 |
| HIPS | 80 | 82 | 85 | 86 | 88 | 89 |
| Size | US | UK / AUS | France | Italy | Japan | Denmark |
|---|---|---|---|---|---|---|
| XS | 0-2 | 6 | 34 | 38 | 5 | 32 |
| S | 2-4 | 8 | 36 | 40 | 7 | 34 |
| M | 6-8 | 10 | 38 | 42 | 9 | 36 |
| L | 8-10 | 12 | 40 | 44 | 11 | 38 |
Description
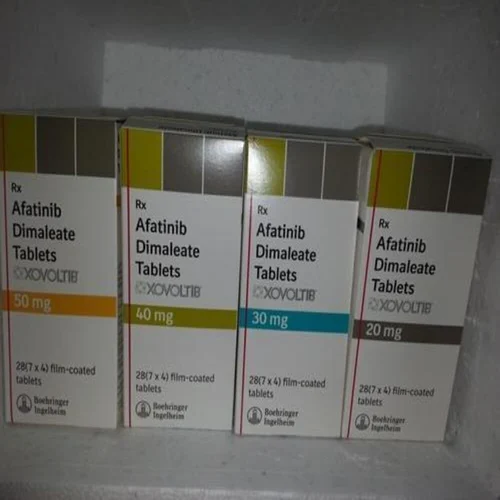
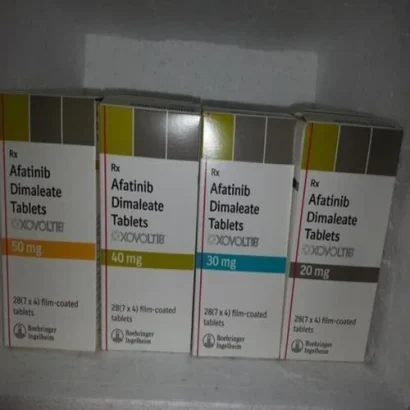
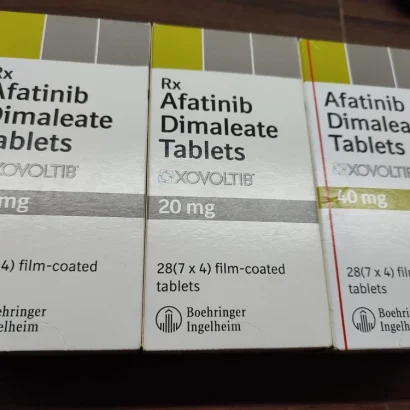
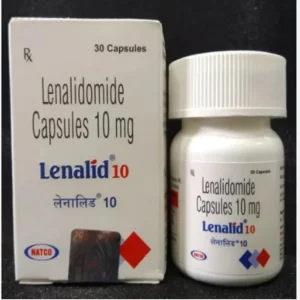
Buy Gilotrif / Xovoltib / Afatinib Dimaleate 20 mg / 30 mg / 40 mg / 50 mg Tablets Online
WHAT IS GILOTRIF?
GILOTRIF is a prescription medicine that is used to treat people with non- small cell lung cancer (NSCLC) that:
has certain (non-resistant) abnormal epidermal growth factor receptor (EGFR) gene(s). Your healthcare provider will perform a test to make sure that GILOTRIF is right for you.
has spread to other parts of the body (metastatic), and
has not been previously treated for metastatic lung cancer
It is not known if GILOTRIF is safe and effective in treating people with lung cancer that has resistant abnormal EGFR genes.
or
is used to treat people with squamous cell lung cancer that:
has spread to other parts of the body, and
has been previously treated with chemotherapy that contains platinum.
It is not known if GILOTRIF is safe and effective in children.
Afatinib is a 4-anilinoquinazoline tyrosine kinase inhibitor in the form of a dimaleate salt available as Boehringer Ingelheim’s brand name Gilotrif Label. For oral use, afatinib tablets are a first line (initial) treatment for patients with metastatic non-small cell lung cancer (NSCLC) with common epidermal growth factor receptor (EGFR) mutations as detected by an FDA-approved test 4. Gilotrif (afatinib) is the first FDA-approved oncology product from Boehringer Ingelheim 4.












There are no reviews yet.